
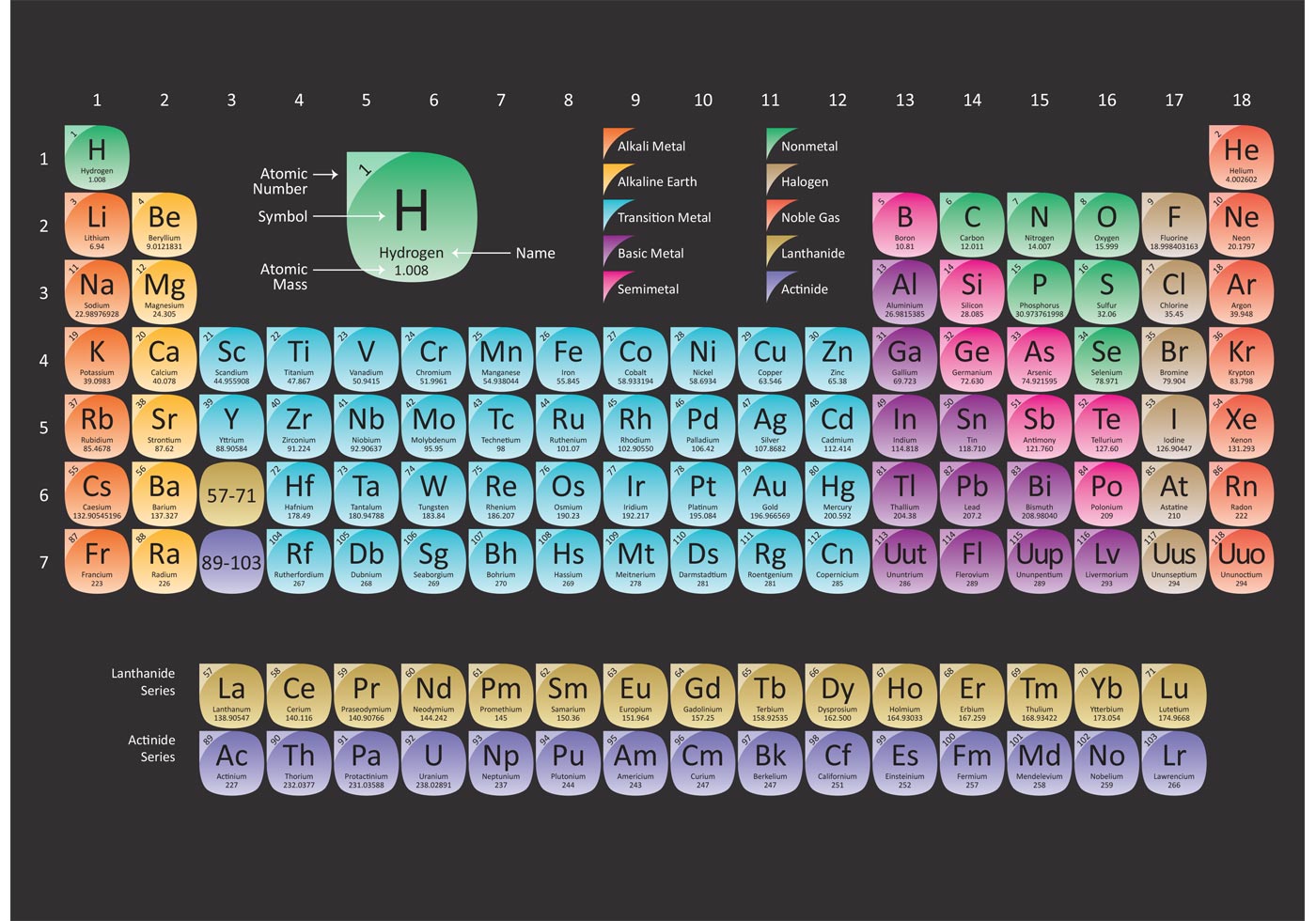
Notes repeat itself in the same way in classification every eighth element in the order of their increasing atomic masses have its property repetition with the first element. Comparatively sa, re, Ga, ma, pa, Dha, ni (in Indian style way), according to his idea like the musical This idea of the periodic table consists of the idea of the law of octaves of the music of West like (do, re, mi, Fa, so, la, ti. Newland’s Octaves law in Periodic Classification of Elements 10th Class

(iv) Several of the known elements were not fit into the triad.Īfter knowing these limitations, the new classification development had occurred. (iii) Only 5 Dobereiner’s triads were identified. (ii) Newly discovered elements are not satisfying the rule. (i) The discovery of new element has ruled out the triads law The limitations of Dobereiner’s triads are as follows:. (In first triad phosphorus mass is average of nitrogen and arsenic mass in second strontium has atomic mass which is a average of calcium and barium) Limitation of Dobereiner Triads in Periodic Classification of Elements Lesson In Dobereiner's triads:- In 1817, a chemist decided to manage element in a table of a triad in which he arranged elements in the order of atomic masses that the atomic mass of the middle element is the average of first and Now let us begin the journey with the ancient to recent one classification.ĭobereiner's triads in Periodic Classification of Elements That’s why modern periodic classification of elements has important As there is on-going discovery of element taking place till now as well. Scientists have done their contribution in this regard with the elements of their time. So, to understand the properties of elements Ziyyara have classified modern periodic classification of elements. And to understand theĬlassification of Elements there physical and chemical properties there is a need of classification. So to understand the chemistry around us first there is a need to understand its basic unit that is element.Īnything can be understood well if there is a proper system. The entire things which are there in universeĪre made up of element. In the form of smallest unit that is atom, and after it comes to molecules and modern periodic classification of elements. Modern Periodic Classification of ElementsĪs we all know that whole universe constitute chemistry in it.

 RSS Feed
RSS Feed
